![]()
Quiz Summary
0 of 40 Questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
0 of 40 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
| Average score |
|
| Your score |
|
Categories
- Not categorized 0%
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
- 22
- 23
- 24
- 25
- 26
- 27
- 28
- 29
- 30
- 31
- 32
- 33
- 34
- 35
- 36
- 37
- 38
- 39
- 40
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 40
1. Question
Which process is used in the preparation of soluble salts?
CorrectIncorrect -
Question 2 of 40
2. Question
Which positive ions are present in aqueous copper(II) sulfate?
CorrectIncorrect -
Question 3 of 40
3. Question
In a titration between an acid (in the burette) and an alkali, you may need to re-use the same
titration flask.
Which is the best procedure for rinsing the flask?CorrectIncorrect -
Question 4 of 40
4. Question
Two containers, one of methane and one of butane, are placed at the same distance from a
naked flame.
Both gases are released at the same time. The methane gas reaches the flame and catches fire
before the butane gas reaches the flame.
Which statement explains this?CorrectIncorrect -
Question 5 of 40
5. Question
Metal X oxidises in air. The formula of the oxide is XO.
X displaces zinc from aqueous zinc nitrate.
Which could be X?CorrectIncorrect -
Question 6 of 40
6. Question
Which is a compound?
CorrectIncorrect -
Question 7 of 40
7. Question
How is a calcium ion, Ca2+, formed from a calcium atom?
CorrectIncorrect -
Question 8 of 40
8. Question
An oxygen atom contains 8 electrons, 8 protons and 10 neutrons.
What is the nucleon number of this atom?CorrectIncorrect -
Question 9 of 40
9. Question
A molecule of sulfuric acid has the structural formula shown.
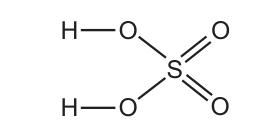
How many electrons are involved in forming all the covalent bonds in one molecule?CorrectIncorrect -
Question 10 of 40
10. Question
A metal consists of a lattice of positive ions in a ‘sea of electrons’.
What happens to the electrons and positive ions in a metal wire when an electric current is
passed through it?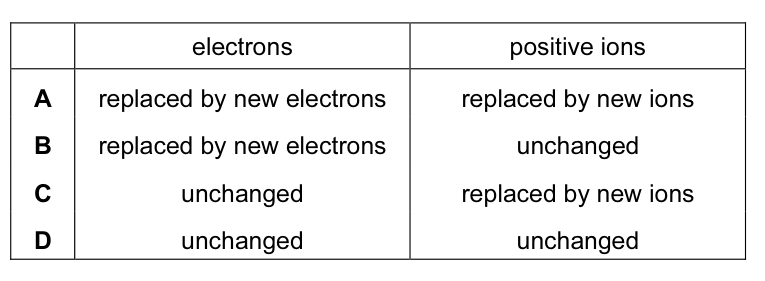 CorrectIncorrect
CorrectIncorrect -
-
Question 11 of 40
11. Question
The equation shown represents the neutralisation of aqueous sodium hydroxide with dilute
sulfuric acid.
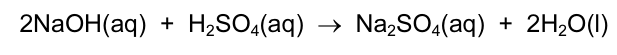
How much sulfuric acid is required to neutralise 100 cm3 of 1.0 mol / dm3 NaOH?CorrectIncorrect -
Question 12 of 40
12. Question
Which change in conditions increases the energy of particles in a reaction?
CorrectIncorrect -
Question 13 of 40
13. Question
Which change is endothermic?
CorrectIncorrect -
Question 14 of 40
14. Question
The enthalpy changes when methane, butane and octane are burned completely in oxygen are
shown below.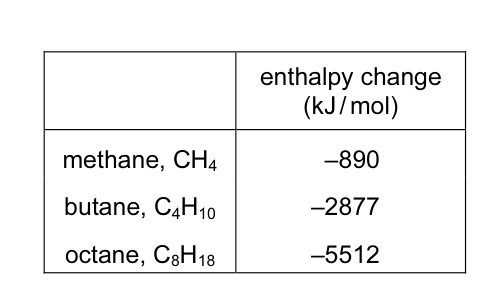
Which are the enthalpy changes when propane and pentane are burned completely in oxygen?
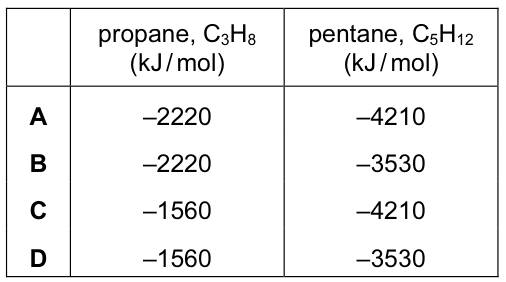 CorrectIncorrect
CorrectIncorrect -
-
Question 15 of 40
15. Question
In the ionic solid zinc phosphide, Zn3P2, what is the formula of the phosphide ion?
CorrectIncorrect -
Question 16 of 40
16. Question
Iron is produced in the blast furnace.
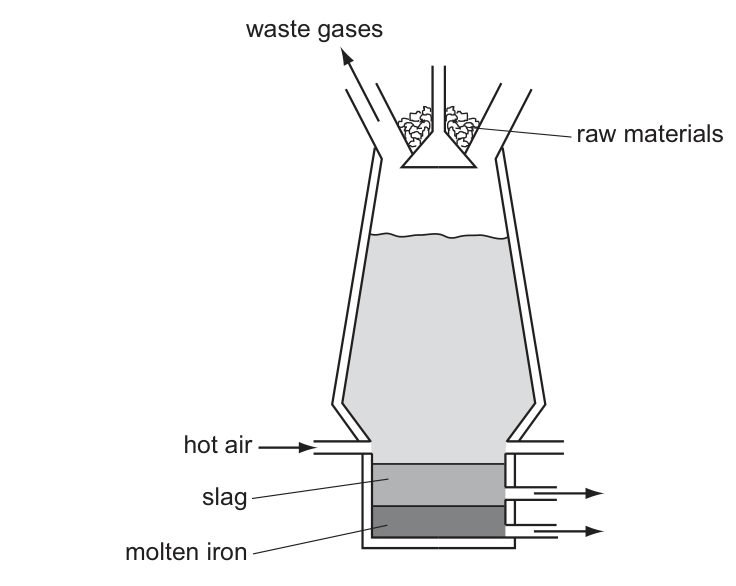
Which statement about this process is correct?
CorrectIncorrect -
Question 17 of 40
17. Question
The energy profile diagram for a reaction is shown.
products
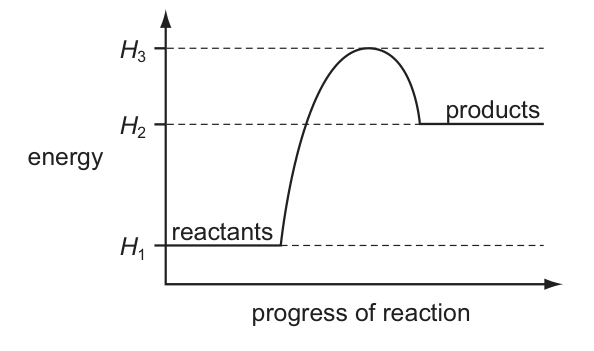
energy
progress of reaction
reactants
Which statement is correct?CorrectIncorrect -
Question 18 of 40
18. Question
In which reaction is nitric acid acting as an oxidising agent?
CorrectIncorrect -
Question 19 of 40
19. Question
Which occurs during the electrolysis of dilute sulfuric acid?
CorrectIncorrect -
Question 20 of 40
20. Question
Methanol is made in industry by a reaction between carbon monoxide and hydrogen.
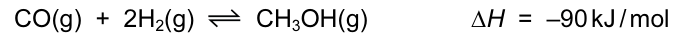
The process is usually carried out at a temperature of 400 °C.
Which row correctly shows the effect on both the position of the equilibrium and on the rate of the
reaction of increasing the temperature to above 400 °C?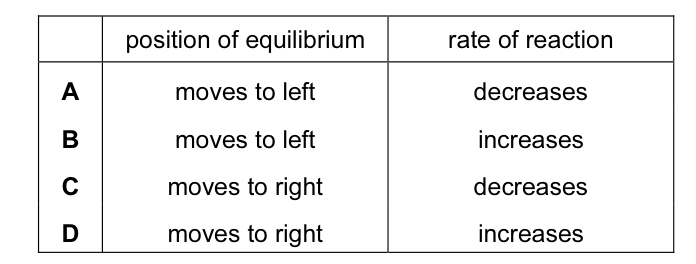 CorrectIncorrect
CorrectIncorrect -
-
Question 21 of 40
21. Question
Which statement about graphite is not correct?
CorrectIncorrect -
Question 22 of 40
22. Question
Ammonium nitrate, NH4NO3, is an artificial fertiliser produced from ammonia.
What is an advantage of using ammonium nitrate as a fertiliser?CorrectIncorrect -
Question 23 of 40
23. Question
Four metals and hydrogen are arranged in order of decreasing reactivity.
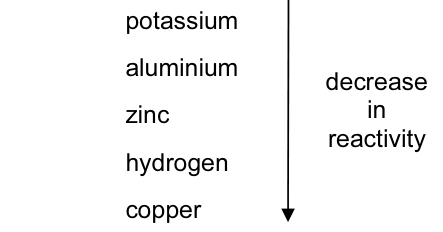
Which statement about these elements is correct?CorrectIncorrect -
Question 24 of 40
24. Question
Which pair of substances reacts to form a salt and water only?
CorrectIncorrect -
Question 25 of 40
25. Question
An element is burned in an excess of oxygen.
Which statement about the oxide formed is always correct?CorrectIncorrect -
Question 26 of 40
26. Question
Which reaction does not involve neutralisation?
CorrectIncorrect -
Question 27 of 40
27. Question
Which element described in the table is a transition metal?
 CorrectIncorrect
CorrectIncorrect -
-
Question 28 of 40
28. Question
Three different elements react by losing electrons. The ions formed all have the electronic
configuration 2,8.
Which statement about these elements is correct?CorrectIncorrect -
Question 29 of 40
29. Question
An alloy of aluminium is used in the construction of aircraft.
Why is pure aluminium never used?CorrectIncorrect -
Question 30 of 40
30. Question
What happens when a strip of silver is immersed in an aqueous solution of copper(II) sulfate?
CorrectIncorrect -
Question 31 of 40
31. Question
The flow chart shows how impure water can be treated to produce drinkable water.
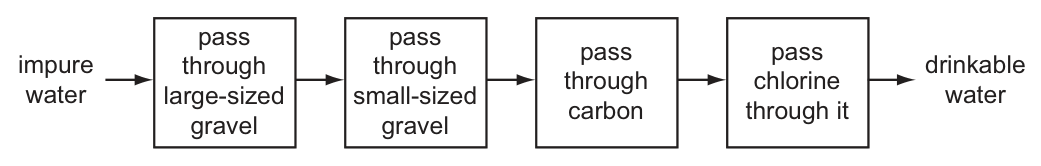
What is not removed from the water by this process?
CorrectIncorrect -
Question 32 of 40
32. Question
Which property of a liquid ester can be used to check its purity before use as a food flavouring?
CorrectIncorrect -
Question 33 of 40
33. Question
Which alcohol will, on oxidation, produce CH3CH2CO2H?
CorrectIncorrect -
Question 34 of 40
34. Question
The diagram shows the structure of an organic acid.
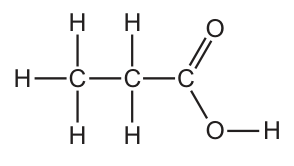
Which row is correct?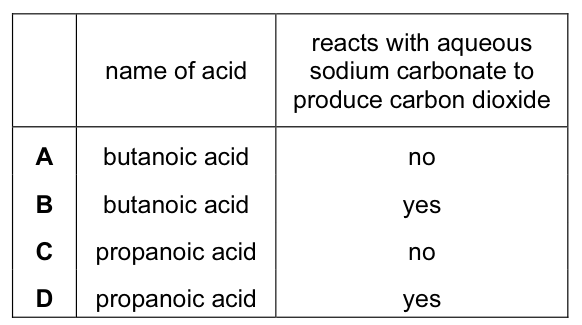 CorrectIncorrect
CorrectIncorrect -
-
Question 35 of 40
35. Question
A carbohydrate such as starch can be represented as shown.
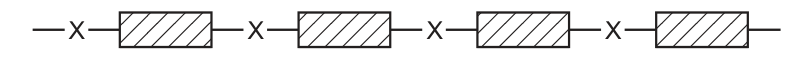
What is X?CorrectIncorrect -
Question 36 of 40
36. Question
P is a polymer that
●
has six carbon atoms in each of the monomers from which it was formed,
●
is not a polyester,
●
was formed using condensation polymerisation.
What is the partial structure of P?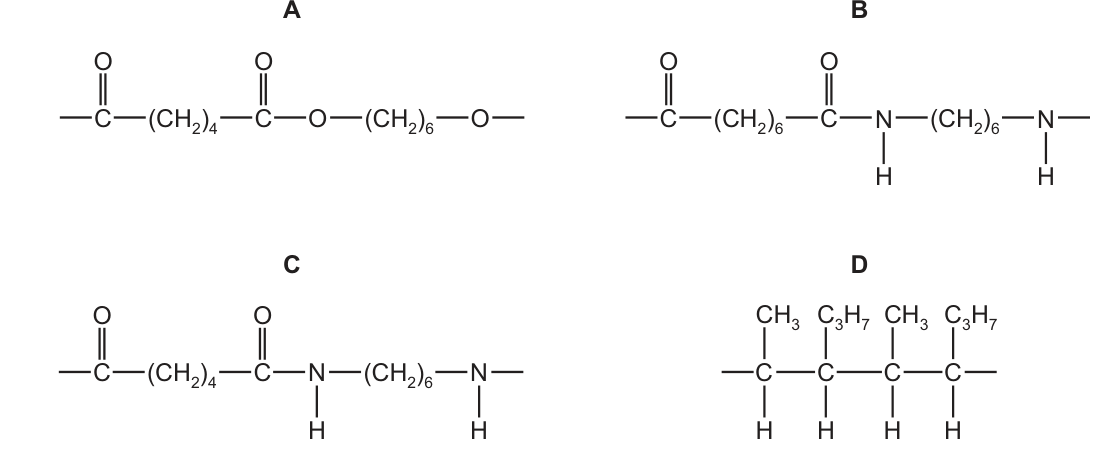 CorrectIncorrect
CorrectIncorrect -
-
Question 37 of 40
37. Question
What are the products of photosynthesis?
CorrectIncorrect -
Question 38 of 40
38. Question
Which diagram shows the structure of an alloy?
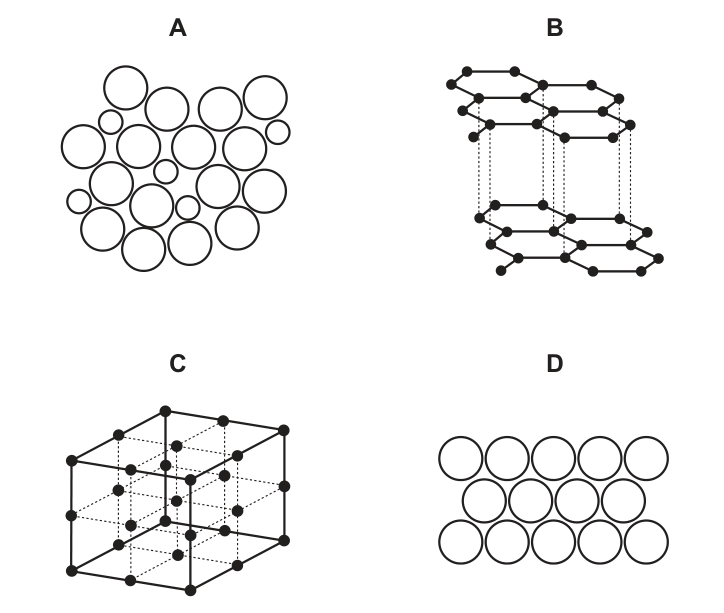 CorrectIncorrect
CorrectIncorrect -
-
Question 39 of 40
39. Question
Hydrogen and nitrogen react to form ammonia.
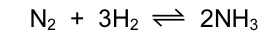
Which statement is correct?
CorrectIncorrect -
Question 40 of 40
40. Question
Which statement about the hydrocarbon C2H4 is not correct?
CorrectIncorrect
